Recommendation: 1
Community structure of heritable viruses in a Drosophila-parasitoids complex
The virome of a Drosophilidae-parasitoid community
Recommended by Ben Longdon based on reviews by 3 anonymous reviewersUnderstanding the factors that shape the virome of a host is key to understanding virus ecology and evolution (Obbard, 2018; French & Holmes, 2020). There is still much to learn about the diversity and distribution of viruses in a host community (Wille et al., 2019; Chen et al., 2023). The viruses of parasitoid wasps are well studied, and their viruses, or integrated viral genes, are known to suppress their insect host’s immune response to enhance parasitoid survival (Herniou et al., 2013; Coffman et al., 2022). Likewise, the insect virome is being increasingly well studied (Shi et al., 2016), with the virome of Drosophila species being particularly well characterised over the best part of the last century (L'Heritier & Teissier, 1937; L'Heritier, 1970; Brun & Plus, 1980; Longdon et al., 2010; Longdon et al., 2011; Longdon et al., 2012; Webster et al., 2015; Webster et al., 2016; Medd et al., 2018; Wallace et al., 2021). However, the viromes of parasitoids and their insect host communities have been less well studied (Leigh et al., 2018; Caldas-Garcia et al., 2023), and the inherent connectivity between parasitoids and their hosts provides an interesting system to study virus host range and cross-species transmission.
Here, Varaldi et al (Varaldi et al., 2024) have examined the viruses associated with a community of nine Drosophilidae hosts and six parasitoids. Using both RNA and DNA sequencing of insects reared for two generations, they selected viruses that are maintained in the lab either via vertical transmission or contamination of rearing medium. From 55 pools of insects they found 53 virus-like sequences, 37 of which were novel. Parasitoids were host to nearly twice as many viruses as their Drosophila hosts, although they note this could be due to differences in the rearing temperatures of the hosts.
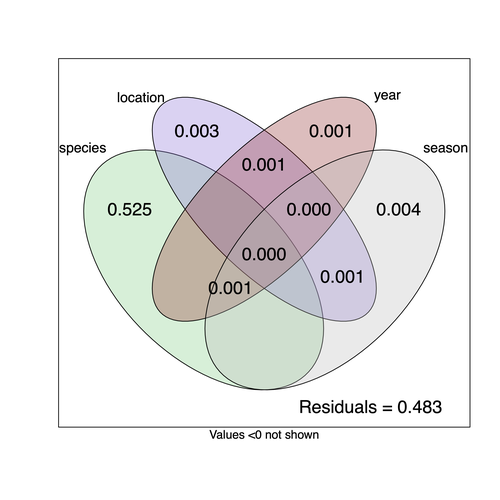
They next quantified if species, year, season, or location played a role in structuring the virome, finding only a significant effect of host species, which explained just over 50% of the variation in virus distribution. No evidence was found of related species sharing more similar virus communities. Although looking at a limited number of species, this suggests that these viruses are not co-speciating or preferentially host switching between closely related species.
Finally, they carried out crosses between lines of the parasitoid Leptopilina heterotoma that were infected and uninfected for a novel Iflavirus found in their sequencing data. They found evidence of high levels of maternal transmission and lower level horizontal transmission between wasp larvae parasitising the same host. No evidence of changes in parasitoid-induced mortality, developmental success or the sex ratio was found in iflavirus-infected parasitoids. Interestingly individuals infected with this RNA virus also contained viral DNA, but this did not appear to be integrated into the wasp genome.
Overall, this work has taken the first steps in examining the community structure of the virome of parasitoids together with their Drosophilidae hosts. This work will not doubt stimulate follow-up studies to explore the evolution and ecology of these novel virus communities.
References
Brun G, Plus N (1980) The viruses of Drosophila. In: The genetics and biology of Drosophila eds Ashburner M & Wright TRF), pp. 625-702. Academic Press, New York.
Caldas-Garcia GB, Santos VC, Fonseca PLC, de Almeida JPP, Costa MA, Aguiar ERGR (2023) The Viromes of Six Ecosystem Service Provider Parasitoid Wasps. Viruses, 15. https://doi.org/10.3390/v15122448
Chen YM, Hu SJ, Lin XD, Tian JH, Lv JX, Wang MR, Luo XQ, Pei YY, Hu RX, Song ZG, Holmes EC, Zhang YZ (2023) Host traits shape virome composition and virus transmission in wild small mammals. Cell, 186, 4662-4675 e4612. https://doi.org/10.1016/j.cell.2023.08.029
Coffman KA, Hankinson QM, Burke GR (2022) A viral mutualist employs posthatch transmission for vertical and horizontal spread among parasitoid wasps. Proceedings of the National Academy of Sciences of the United States of America, 119. https://doi.org/10.1073/pnas.2120048119
French RK, Holmes EC (2020) An Ecosystems Perspective on Virus Evolution and Emergence. Trends in Microbiology, 28, 165-175. https://doi.org/10.1016/j.tim.2019.10.010
Herniou EA, Huguet E, Thézé J, Bézier A, Periquet G, Drezen JM (2013) When parasitic wasps hijacked viruses: genomic and functional evolution of polydnaviruses. Philosophical Transactions of the Royal Society B-Biological Sciences, 368. https://doi.org/10.1098/rstb.2013.0051
L'Heritier PH (1970) Drosophila viruses and their role as evolutionary factors. Evolutionary Biology, 4, 185-209
L'Heritier PH, Teissier G (1937) Une anomalie physiologique héréditaire chez la Drosophile. C.R. Acad. Sci. Paris, 231, 192-194
Leigh BA, Bordenstein SR, Brooks AW, Mikaelyan A, Bordenstein SR (2018) Finer-Scale Phylosymbiosis: Insights from Insect Viromes. Msystems, 3. https://doi.org/10.1128/mSystems.00131-18
Longdon B, Obbard DJ, Jiggins FM (2010) Sigma viruses from three species of Drosophila form a major new clade in the rhabdovirus phylogeny. Proceedings of the Royal Society B, 277, 35-44.
https://doi.org/10.1098/rspb.2009.1472
Longdon B, Wilfert L, Jiggins FM (2012) The Sigma Viruses of Drosophila. Caister Academic Press, Norfolk, UK.
Longdon B, Wilfert L, Osei-Poku J, Cagney H, Obbard DJ, Jiggins FM (2011) Host switching by a vertically-transmitted rhabdovirus in Drosophila. Biology Letters, 7, 747-750.
https://doi.org/10.1098/rsbl.2011.0160
Medd NC, Fellous S, Waldron FM, Xuereb A, Nakai M, Cross JV, Obbard DJ (2018) The virome of Drosophila suzukii, an invasive pest of soft fruit. Virus Evol, 4, vey009.
https://doi.org/10.1093/ve/vey009
Obbard DJ (2018) Expansion of the metazoan virosphere: progress, pitfalls, and prospects. Curr Opin Virol, 31, 17-23. https://doi.org/10.1016/j.coviro.2018.08.008
Shi M, Lin XD, Tian JH, Chen LJ, Chen X, Li CX, Qin XC, Li J, Cao JP, Eden JS, Buchmann J, Wang W, Xu J, Holmes EC, Zhang YZ (2016) Redefining the invertebrate RNA virosphere. Nature. https://doi.org/10.1038/nature20167
Varaldi J, Lepetit D, Burlet N, Faber C, Baretje B, Allemand R (2024) Community structure of heritable viruses in a Drosophila-parasitoids complex. bioRxiv, 2023.2007.2029.551099, ver.3, peer-reviewed and recommended by Peer Community in Evolutionary Biology. https://doi.org/10.1101/2023.07.29.551099
Wallace MA, Coffman KA, Gilbert C, Ravindran S, Albery GF, Abbott J, Argyridou E, Bellosta P, Betancourt AJ, Colinet H, Eric K, Glaser-Schmitt A, Grath S, Jelic M, Kankare M, Kozeretska I, Loeschcke V, Montchamp-Moreau C, Ometto L, Onder BS, Orengo DJ, Parsch J, Pascual M, Patenkovic A, Puerma E, Ritchie MG, Rota-Stabelli O, Schou MF, Serga SV, Stamenkovic-Radak M, Tanaskovic M, Veselinovic MS, Vieira J, Vieira CP, Kapun M, Flatt T, Gonzalez J, Staubach F, Obbard DJ (2021) The discovery, distribution, and diversity of DNA viruses associated with Drosophila melanogaster in Europe. Virus Evol, 7, veab031. https://doi.org/10.1093/ve/veab031
Webster CL, Longdon B, Lewis SH, Obbard DJ (2016) Twenty-Five New Viruses Associated with the Drosophilidae (Diptera). Evol Bioinform Online, 12, 13-25. https://doi.org/10.4137/EBO.S39454
Webster CL, Waldron FM, Robertson S, Crowson D, Ferrai G, Quintana JF, Brouqui JM, Bayne EH, Longdon B, Buck AH, Lazzaro BP, Akorli J, Haddrill PR, Obbard DJ (2015) The discovery, distribution and evolution of viruses associated with Drosophila melanogaster. Plos Biology, 13(7): e1002210. https://doi.org/10.1371/journal.pbio.1002210
Wille M, Shi M, Klaassen M, Hurt AC, Holmes EC (2019) Virome heterogeneity and connectivity in waterfowl and shorebird communities. ISME J, 13, 2603-2616. https://doi.org/10.1038/s41396-019-0458-0

