
Hooked on Wolbachia
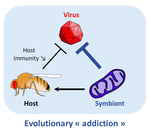
Addicted? Reduced host resistance in populations with defensive symbionts
Abstract
Rivero, A. and Kremer, N. (2017) Hooked on Wolbachia. Peer Community in Evolutionary Biology, 100004. https://doi.org/10.24072/pci.evolbiol.100004
Recommendation
This very nice paper by Martinez et al. [1] provides further evidence, if further evidence was needed, of the extent to which heritable microorganisms run the evolutionary show.
Wolbachia is an ubiquitous endosymbiont of arthropods who has been recently shown to protect its hosts against viral infections. Here, Martinez et al. are able to show that this multifaceted heritable symbiont weakens selective pressures induced by viruses on host immune genes. In a series of very elegant experiments, Wolbachia-infected and Wolbachia-free populations of D. melanogaster were exposed to Drosophila C virus (a natural, and highly virulent Drosophila pathogen). At the end of a 9-generation artificial selection protocol with DCV, resistance against DCV increased in flies, both in the presence and absence of Wolbachia. Wolbachia-infected flies were still substantially more resistant to DCV viruses than their Wolbachia-free counterparts. Crucially, however, the frequency of the pastrel resistant allele (a key immune gene for DCV resistance) was significantly lower in the Wolbachia-infected lines. As a consequence, when the DCV-evolved lines were treated with antibiotics to cure them from the bacterial infection, the lines who had evolved with Wolbachia tended to be more susceptible to the virus than their uninfected counterparts.
In other words, infection by protective heritable symbionts can affect how selection acts on the host's nuclear-based resistance, effectively rendering it dependent on its symbiont for the fight against pathogens.
But the interest of these results may not be simply academic. The protective qualities of Wolbachia against a range of pathogens have opened up the exciting possibility of transferring these bacteria to mosquito vectors of key human diseases such as dengue or malaria. The long term evolutionary potential for these novel Wolbachia-host interactions has, however, been little explored. Either the Wolbachia, the pathogen or, as shown here, the host, could evolve in more or less predictable ways. There is, for example, evidence showing that in novel hosts Wolbachia evolves rapidly and tends to gradually lose its virulence. If the lost virulence was to result in a decrease in their pathogen defensive qualities, the mosquito, having lost the efficiency of its conventional immune defences, could end up being more vulnerable to infection than before the Wolbachia introduction. Martinez et al.'s paper is a nice example of how investigating the evolutionary potential of such Wolbachia-host-pathogen interactions can be hugely informative as to the long term prospects of these new control methods.
Reference
[1] Martinez J, Cogni R, Cao C, Smith S, Illingworth CJR & Jiggins FM. 2016. Addicted? Reduced host resistance in populations with defensive symbionts. Proceedings of the Royal Society of London B 283:20160778. doi: 10.1098/rspb.2016.0778










