
Cancer and loneliness in Drosophila
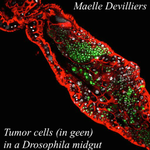
An interaction between cancer progression and social environment in Drosophila
Abstract
Recommendation: posted 20 September 2017, validated 20 September 2017
Rivero, A. (2017) Cancer and loneliness in Drosophila. Peer Community in Evolutionary Biology, 100030. https://doi.org/10.24072/pci.evolbiol.100030
Recommendation
Drosophila flies may not be perceived as a quintessentially social animal, particularly when compared to their eusocial hymenopteran cousins. Although they have no parental care, division of labour or subfertile caste, fruit flies nevertheless exhibit an array of social phenotypes that are potentially comparable to those of their highly social relatives. In the wild, Drosophila adults cluster around food resources where courtship, mating activity and oviposition occur. Recent work has shown not only that social interactions in these clusters condition many aspects of the behaviour and physiology of the flies [1] but also, and perhaps more unexpectedly, that social isolation has a negative impact on their fitness [2].
Many studies in humans point to the role of social isolation as a source of stress that can induce and accelerate disease progression. The ultimate proof of the connection between social interaction and disease is however mired in confounding variables and alternative explanations so the subject, though crucial, remains controversial. With a series of elegant experiments using Drosophila flies that develop an inducible form of intestinal cancer, Dawson et al [3] show that cancer progresses more rapidly in flies maintained in isolation than in flies maintained with other cancerous flies. Further, cancerous flies kept with non-cancerous flies, fare just as badly as when kept alone. Their experiments suggest that this is due to the combined effect of healthy flies avoiding contact with cancerous flies (even though this is a non-contagious disease), and of cancerous flies having higher quality interactions with other cancerous flies than with healthy ones. Perceived isolation is therefore as pernicious as real isolation when it comes to cancer progression in these flies. Like all good research, this study opens up as many questions as it answers, in particular the why and wherefores of the flies’ extraordinary social behaviour in the face of disease.
References
[1] Camiletti AL and Thompson GJ. 2016. Drosophila as a genetically tractable model for social insect behavior. Frontiers in Ecology and Evolution, 4: 40. doi: 10.3389/fevo.2016.00040
[2] Ruan H and Wu C-F. 2008. Social interaction-mediated lifespan extension of Drosophila Cu/Zn superoxide dismutase mutants. Proceedings of the National Academy of Sciences, USA, 105: 7506-7510. doi: 10.1073/pnas.0711127105
[3] Dawson E, Bailly T, Dos Santos J, Moreno C, Devilliers M, Maroni B, Sueur C, Casali A, Ujvari B, Thomas F, Montagne J, Mery F. 2017. An interaction between cancer progression and social environment in Drosophila. BiorXiv, 143560, ver. 3 of 19th September 2017. doi: 10.1101/143560
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article. The authors declared that they comply with the PCI rule of having no financial conflicts of interest in relation to the content of the article.
Evaluation round #2
DOI or URL of the preprint: 10.1101/143560
Version of the preprint: 2
Author's Reply, 01 Sep 2017
Decision by Ana Rivero, posted 01 Sep 2017
Dear authors,
Thank you for the replies to our comments and suggestions. There are still some issues that the reviewers feel are important to improve the quality of the paper and that were not fully addressed in your earlier replies.
If you choose to answer to these points, and to facilitate our work, I would ask you to please indicate in your replies where in the manuscript the changes have been made (provide line numbers)
REVIEWER 1
Results
Page 7 (Social environment choice)
(9) (second para.) I see how what you are saying in this paragraph fits well with what I see in Figure 3, however I got confused by the stats. For example, it looks like the target fly effect is only significant 7 days post induction, so I find it surprising that you have a significant target fly main effect but a not significant target fly*age interaction. As I read on, I’m not even sure whether what is being tested is the difference between control and cancerous targets, or between observed vs expected (random) choice. Where does this second chi-square value for age come from? A bit more info about how was this analysis done would help understand.
AUTHOR’s REPLY
We now provide more information on the way the analyses were done. We apologize but found an error in the statistical analysis of the dual choice (age is in fact strongly significant, no change in the results of the other factors). We observed a general decrease in preference for the cancerous stimulus group, but we could detect such decrease only in cancerous target flies. This may explain the lack of significant interaction target fly*age.
RECOMMENDER REPLY #1
The analyses of the choice experiments still lack clarity. These issues have also been raised by REVIEWER 2, please see below.
(19) Was the “tube” effect taken into account in the model? i.e. is this a mixed model with treatment as a fixed factor and tube as a random one?
AUTHOR’s REPLY
Because in the heterogeneous environment only one cancerous fly was present, we grouped fly guts randomly from different tubes (this is now mentioned in the manuscript). Thus tube was not a random factor and only treatment was included as a fixed factor.
RECOMMENDER’s REPLY #2
Does this mean that for the “alone” and the “heterogeneous” treatments the % of cancerous flies is given for an individual cancerous fly and that for the “homogeneous” treatment it is the mean for the 8 cancerous flies in the tube? But then what does it mean that you “grouped fly guts randomly from different tubes”? (and where in the manuscript is this point clarified? Please provide line numbers).
REVIEWER 2
Major comments
- I am confused with the statistical analysis in the choice experiments. The authors show the results of a logistic regression in the text, where they ask the questions whether age, cancerous state/stimulus, or the interaction between them affects choice. They are thus comparing different groups in their choice, but they do not directly test for one group whether they are more attracted to a certain cage over the other (just differences between groups). This is more or less tested in individual tests for deviation of random choice (0.5) with asterisks in figures 3-4, however, the method of this testing is not described but seems to be performed for each individual point separately and p-values should thus be adjusted for multiple testing. Was this done? I suggest the authors to have the principle analysis be done on whether cancerous flies are attracted to a certain social group, a secondary analysis would be whether there are differences between cancerous vs control and age of the fly.
AUTHOR’s REPLY
Stars on figure 3 and 4 represent significant deviation from random choice calculated for each line and age. We disagree with the necessity of doing the adjustment for multiple testing as we are not comparing the treatments among them in this analysis (compared to figure 1 and 2).
We believe that this representation allows the reader to see at which age and state there is a significant effect and can conclude that there is for example aversion of the cancerous flies by the control ones when tumours are well developed. We clarified each analysis done and detail the statistics.
RECOMMENDER REPLY #3
3.1. We agree that this is a useful way of looking at the data, but the question asked by reviewer to was how was the significant deviation from random choice tested. Please state what statistical test was used.
3.2. Contrary to what the authors state, the adjustment for multiple testing is required irrespective of whether you’re comparing treatments with each other or, as is the case here, comparing observed vs expected (random) 6 different times (one for each day x fly state combination). You could easily do a (back of the envelope) Bonferroni correction by dividing your critical alpha (0.05) by the number of tests (0.05/6 = 0.008). Thus, you would then only consider tests with p<0.008 (or to round it up p<0.01) to be significant.
- P7, paragr3: “This was especially pronounced when flies were young i.e. at the very beginning of the tumor development”. However, the interaction between age and target fly is not significant so this is not a significant effect.
AUTHOR’s REPLY
We made this clear that here we were not comparing cancerous vs control
RECOMMENDER REPLY #4
Not that clear, actually. The analyses in Lines 178-179 seem to be comparing control vs cancerous (this is the “target fly” effect, right?). As stated by the reviewer, since the interaction age x target fly is not significant, stating that “this was especially pronounced when flies were young” is, statistically speaking, incorrect (even if there seems to be a trend in that direction).
- P7, paragr3: “However, at later … P<10-3).” I believe this p-value must be based on the individual datapoint analysis that is not described. This becomes confusing because you first report a non-significant interaction (see point above), but here you don´t talk about a difference between groups, but a difference from random choice. Please rewrite the results so these distinctions become clearer.
AUTHOR’s REPLY
We hope this is now clarified
RECOMMENDER REPLY #5
Not really. Please clarify what analyses have been done (i.e. what is it exactly that you’re testing?) in lines 180-181 and lines 183-184 and how they differ from the analysis reported in lines 178-179. I’m guessing 178-179 is cancerous vs target, while the other analyses test for a departure from randomness for cancerous (180 181) and control (183-184) flies. If so, this is far from clear from the text. Please see also #6 regarding clarifying the significance of the intercept.
- P7, paragr4: “Cancerous flies showed … P=0.44”, same point as above, you report the nonsignificant result but mention a significant effect. Report statistical analysis.
AUTHOR’s REPLY
We apologize, the significance of attraction towards the social stimulus could only be understood by mentioning the p value of the intercept. It has been added.
RECOMMENDER REPLY #6
6.1 Thanks for the clarification. As an intercept can be interpreted in many different ways depending on the type of model, the fact that in a logistic regression a significant intercept indicates a departure from randomness (which may or may not be explained by the explanatory variables) needs to be clearly stated here (Line 454-455 is lost in the depths of the m&m and does not suffice). If possible, please provide a reference.
6.2 I take it the individual departure from each of the treatment combinations (indicated by the stars in Figure 4) was done separately? If so please state how (as per #3.1 and #3.2).
6.3 Why are there no stats comparing Cancerous vs Control target flies?
- In the concluding paragraph of the discussion there is a referral to the contribution of this study to the evolutionary ecology of cancer, it would be great if the authors could expand a few sentences on this. What are the evolutionary benefits? Could such behavior be adaptive for cancer or is it an unintended consequence of a non-specific infection avoiding behavior? The tumor cells in this study do not impact fitness, could this bias any conclusions drawn from this study?
AUTHOR’s REPLY
We modified the discussion accordingly to this comment
RECOMMENDER REPLY #7
I have failed to see any substantial change to the discussion. Could you please be more precise as to how and where (line numbers) these changes have been made?
Minor comments:
- P8, paragr4 “Even if not… with being sick”: if this is a general response, shouldn´t cancerous flies also avoid other cancerous flies (which they don´t)? If it is a general response, they may want to avoid flies with contagious infection despite themselves having cancerous cells. Please discuss.
AUTHOR’s REPLY
Even if we have not observed clear ‘avoidance’ of the cancerous flies by other cancerous ones we still see a decrease in preference. We believe that there could be a balance between avoidance of the potentially contagious individual and attraction of individual of the same type. This balance may vary with cancer progression.
RECOMMENDER REPLY #8
Please clarify whether this clarification has been included in the manuscript and if so where.
Evaluation round #1
DOI or URL of the preprint: 10.1101/143560
Version of the preprint: 1
Author's Reply, 19 Jul 2017
Decision by Ana Rivero, posted 15 Jul 2017
Dear Authors,
This is a very interesting article that addresses an important issue on the correlation between cancer and social environment, using a relevant experimental system. However, both referees have raised issues with the statistical analyses: they lack clarity and at times they seem to be at odds with what it's written in the text. Would be happy to recommend provided the statistical analyses are clarified and all the individual points raised by the referees (statistical, or otherwise) addressed.
Best
Ana
Reviewed by Ana Rivero, 10 Jul 2017
Review Dawson et al The relationship between cancer progression and social environment in Drosophila
This is an original manuscript looking at the effects of sociality on non-infection disease progression and vice-versa. Please see below for a detail list of comments and suggestions which I hope will increase the clarity of the manuscript (because the lack of line numbers I found it easier to list them in order of appearance, rather than importance). The short format makes for a slightly frustrating read, as I found myself wanting to know much more about these tumours, and about the potential mechanistic and adaptive explanations behind these rather sophisticated behaviours (cancerous Drosophila flies that prefer to mix with other cancerous flies rather than healthy ones - wow). Other than that, and as you will see, my main comments relate to the statistical analyses, which I think could be clarified pretty much throughout.
Introduction
Page 5 (first para.), (1) Previous examples of the correlation between cancer progression and social isolation have been described in intrinsically social mammals such as humans and mice. I have a hard time thinking about Drosophila as a stereotypically social animal, perhaps a few lines about adult Drosophila social behaviour would help to better understand why this is a good model for addressing this issue. (2) To understand the adaptive nature of Drosophila faced with cancerous vs non cancerous flies I also feel the need to know more about these “intestinal-like cancers” (which is a peculiar way to call them – do you mean “intestinal cancer-like tumours?”). Do they occur naturally or are they a laboratory construct? What are their fitness effects on the flies (if any?). It seems surprising that these tumours have no effect on fly performance or longevity (page 24)– so what do they do? And why would you expect Drosophila to have evolved adaptive strategies to deal with them?
Results
Page 5 (Biological model) (3) What is MARCM and what does it mean that “The flies contained […] MARCM clones”?
Page 6 (first para.) (4) Substitute “bred in tubes” for “kept in tubes” (there was no breeding involved) (5) The last couple of sentences “After 21 days…” and “More surprisingly…” require a statistical test (in the form of a post-hoc contrast – you can do this by e.g. lumping together the alone and heterogeneous treatment and checking whether there’s a significant change in deviance in the model)
Page 6 (Social interactions para.) (6) A general comment I have throughout the paper is that there is very little information about the statistical analyses. It would help if the Methods had a Statistical Analysis section detailing how the analyses were made (what program? How were the models built? Were they simplified? that kind of thing). Here, for example, you have 3 different F and p values associated to the analyses in figures 2B. Are these 3 different statistical analyses (I hope not) or post-hoc contrasts after fitting a full mixed model (to account for the tube effect) with group composition and fly state (and their interaction) as fixed explanatory variables? This seems to be the appropriate way to do this analysis. (7) Same goes for analyses of data in 2C. (8) I’m also wandering how was the data handled, particularly in the heterogeneous group, where you have a higher replication for the control flies (n=7 per tube) than for the cancerous flies (n=1). Unless I’m mistaken, you do not seem to mention how many true replications (tubes) of each there are.
Page 7 (Social environment choice)
(9) (second para.) I see how what you are saying in this paragraph fits well with what I see in Figure 3, however I got confused by the stats. For example, it looks like the target fly effect is only significant 7 days post induction, so I find it surprising that you have a significant target fly main effect but a not significant target fly*age interaction. As I read on, I’m not even sure whether what is being tested is the difference between control and cancerous targets, or between observed vs expected (random) choice. Where does this second chi-square value for age come from? A bit more info about how was this analysis done would help understand.
(10) Figure 4 (there is no Figure 5) could be clearer: I would change X axis label to “Stimulus flies (days post-induction)” and add “Target flies” to the cancerous and control labels. I would also call these latter ones “empty” rather than control.
(11) The exact same comments regarding the stats as for the dual experiment apply here.
Discussion
Page 8 (12) (second para.) I do not understand why, if social isolation is the sole issue, cancerous flies do better in the presence of other cancerous flies rather than in the presence of healthy flies. I find this result very interesting but also very perplexing. Any thoughts? (13) You also suggest (page 9) that the attraction of cancerous flies to other cancerous flies may be an adaptive strategy aimed at reducing cancer progression. To be convinced by these adaptive explanations, I would need to know more about the conditions under which these tumours originated (see comments on Intro).
Methods
Page 12 (last para) (14) The people that’ve named these Drosophila lines seem to have some serious psychological issues (!). Because of all the colons and semicolons it took me a while to understand this. You may want to cater for people like me by modifying the initial sentence like thus: “We used two different Drosophila lines: a cancerous line (yw, HS-flp….) and a control line (yw, HS-flp….) which were balanced over co-segregating….”. Please also explain what does this last part of the sentence mean (“balanced over…”). (15) “In all experiments flies were…” this seems to be a repeat of what has already been said (or else the subtlety was lost on me).
Page 13 (first para) (16) This would indeed have been the perfect control for your experiment, as otherwise the temperature treatment is confounded with the Apc-Ras mutation. To what extent could your results be simply explained by the heat shock instead of the tumour? A quick check of the literature shows that hsps can have multiple pleiotropic effects in the organism, including on fly behavior. This is not discussed in the manuscript, but I think it should. Would an alternative have been to have a heat-shocked control line in the behavioural experiments?
Page 13 (social breeding) (17) It may be a good idea to call this “social environment” as per the Results section (18) How many food tubes were there for each of the 3 treatments? (19) Was the “tube” effect taken into account in the model? i.e. is this a mixed model with treatment as a fixed factor and tube as a random one?
Page 14 (Social environment choice) (20) I had to read the experimental set up a few times to understand it. To avoid confusion between the different types of cages, it may be a good idea to refer to the outer cage as a “plastic box”.
Supplementary Information
Page 24
(21) “and locomotor” …activity?
(22) How many groups of cancerous / control females were used for each of the 3 post-induction treatments?
(23) Please tell us what was used as the random variable (tube?) in the mixed model.
(24) You seem to have three fixed parameters (instead of 2): fly state, time and age. Not sure what you mean by “age” though (is this the post-induction treatment? i.e. 7, 14 and 21 days? – if so it may be a good idea to keep terminology constant)
(25) “no difference between cancerous or control flies could be observed at any age (p>0.5 for each age tested)” – how were these multiple tests done? Are these post-hoc contrasts? This sentence is misleading (as it sounds like you have done separate statistical tests for each age, which would not be a good idea). Stating that you do not have a significant interaction between fly state and age (if this indeed the case), would obviate the need to do either multiple tests or contrasts.










