Latest recommendations
| Id | Title * | Authors * | Abstract * | Picture * | Thematic fields * | Recommender | Reviewers | Submission date | |
|---|---|---|---|---|---|---|---|---|---|
07 Feb 2025
Discordant population structure inferred from male- and female-type mtDNAs from Macoma balthica, a bivalve species characterized by doubly uniparental inheritance of mitochondriaSabrina Le Cam, Brémaud Julie, Vanessa Becquet, Valerie Huet, Pascale Garcia, Amélia Viricel, Sophie Breton, Eric Pante https://doi.org/10.1101/2022.02.28.479517Unraveling the Complexities of Mitochondrial Inheritance in Macoma balthica: Insights from Doubly Uniparental InheritanceRecommended by Rita Castilho based on reviews by Risto Väinölä and John WaresThe study by Le Cam and colleagues, entitled “Discordant population structure inferred from male- and female-type mtDNAs from Macoma balthica, a bivalve species characterized by doubly uniparental inheritance of mitochondria”, provides a fascinating exploration of the genetic structure and evolutionary dynamics of the Baltic tellin, Macoma balthica, a bivalve species exhibiting doubly uniparental inheritance (DUI) of mitochondria. This work is a significant contribution to the field of evolutionary biology, particularly in understanding how sex-specific mitochondrial inheritance can shape population structure and genetic diversity in marine organisms. DUI is a remarkable exception to the typical maternal inheritance of mitochondria in metazoans, where both males and females can transmit their mitochondria, but through different routes. In species with DUI, females pass on their mitochondria to all offspring, while males transmit their mitochondria exclusively to their sons. This system results in males being heteroplasmic, carrying both maternal (F-type) and paternal (M-type) mitochondrial DNA (mtDNA). The study leverages this unique inheritance pattern to investigate the genetic diversity, divergence, and population structure of M. balthica across its distribution range, from the North Sea to the Gironde Estuary in Southern France. One of the most striking findings of this study is the discordant population structure inferred from the male- and female-type mtDNAs. The authors sequenced the cox1 gene from both F-type and M-type mtDNA in 302 male individuals across 14 sampling sites. They found that the genetic differentiation between northern and southern populations was nearly three times higher for the M-type mtDNA compared to the F-type mtDNA. This discrepancy was further highlighted by the geographic localization of the strongest genetic break, which differed significantly between the two markers. For the F-type mtDNA, the break was located at the Finistère Peninsula, while for the M-type mtDNA, it was found at the Cotentin Peninsula, approximately 250 km apart. The authors propose several explanations for these differences, including a higher mutation rate, relaxed negative selection, and variations in effective population sizes for the M-type mtDNA. These factors could contribute to the observed divergence in genetic structure between the two mitochondrial types. Additionally, the study suggests that mito-nuclear genetic incompatibilities, arising from the interaction between mitochondrial and nuclear genes involved in oxidative phosphorylation and ATP production, could play a role in maintaining these genetic barriers. The study also provides valuable insights into the phylogeographic history of M. balthica. The divergence times estimated for the F-type and M-type mtDNA clades suggest that the split between the northern and southern populations occurred before the last glacial maximum (LGM). This finding supports a scenario of pre-LGM vicariance rather than post-glacial primary intergradation. The authors' use of net divergence to estimate the timing of cladogenesis events within the Macoma species complex adds a robust temporal dimension to their phylogeographic analysis. Another intriguing aspect of the study is the evidence of asymmetric introgression and hybrid zone dynamics. The authors observed that the genetic clines for the F-type and M-type mtDNAs were not only discordant in their geographic locations but also in their widths. The cline for the M-type mtDNA was significantly narrower than that for the F-type mtDNA, suggesting different selective pressures or migration balances acting on the two mitochondrial types. This finding raises important questions about the role of sex-specific selection and gene flow in shaping the genetic structure of populations with DUI. The study also highlights the importance of considering sex-specific genetic markers in population genetic studies. One of the most intriguing aspects is the implication that M-type mtDNA may be evolving under different constraints than its female counterpart. The authors found higher nucleotide diversity and net divergence in M-type sequences suggestive of a relaxed selective regime or an increased mutation rate, which aligns with previous studies on DUI species. Given that M-type mitochondria function predominantly in sperm cells, the potential for oxidative damage, reduced purifying selection, and a higher mutation rate could explain these patterns. More broadly, this finding reinforces the idea that mitochondrial genomes, though typically constrained by purifying selection, can evolve along sex-specific trajectories when their inheritance is decoupled from maternal transmission.
References Sabrina Le Cam, Brémaud Julie, Vanessa Becquet, Valerie Huet, Pascale Garcia, Amélia Viricel, Sophie Breton, Eric Pante (2025) Discordant population structure inferred from male- and female-type mtDNAs from Macoma balthica, a bivalve species characterized by doubly uniparental inheritance of mitochondria. bioRxiv, ver.4 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.1101/2022.02.28.479517 | Discordant population structure inferred from male- and female-type mtDNAs from Macoma balthica, a bivalve species characterized by doubly uniparental inheritance of mitochondria | Sabrina Le Cam, Brémaud Julie, Vanessa Becquet, Valerie Huet, Pascale Garcia, Amélia Viricel, Sophie Breton, Eric Pante | <p> Doubly Uniparental Inheritance (DUI) of mitochondria is a remarkable exception to the Strictly Maternal Inheritance (SMI) in metazoans. In species characterized by DUI --almost exclusively gonochoric bivalve mollusks-- females (F) transmi... | Phylogeography & Biogeography, Population Genetics / Genomics | Rita Castilho | 2022-03-03 10:22:01 | View | ||
31 Jan 2025
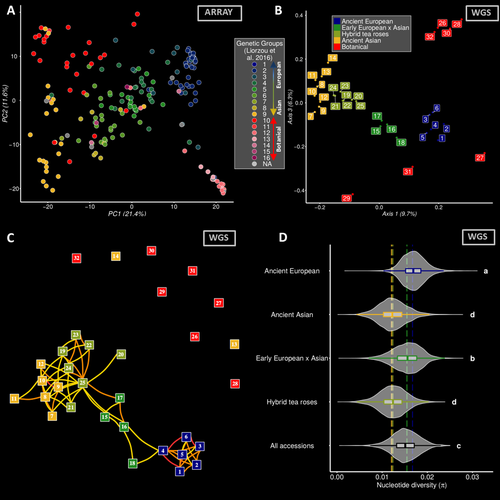
Dark side of the honeymoon: reconstructing the Asian x European rose breeding history through the lens of genomicsThibault Leroy, Elise Albert, Tatiana Thouroude, Sylvie Baudino, Jean-Claude Caissard, Annie Chastellier, Jerome Chameau, Julien Jeauffre, Therese Loubert, Saretta Nindya Paramita, Alix Pernet, Vanessa Soufflet-Freslon, Cristiana Oghina-Pavie, Fabrice Foucher, Laurence Hibrand-Saint Oyant, Jeremy Clotault https://doi.org/10.1101/2023.06.22.546162A genomic view on the history of rose garden breeding in EuropeRecommended by mathieu gautier based on reviews by Pierre Nouhaud, Vincent Segura and 1 anonymous reviewerRoses, a symbol of love and beauty, have a rich history of cultivation that spans millennia. While their aesthetic appeal has remained a constant, the genetic changes underlying their domestication and breeding remain intriguing. The manuscript titled "Dark side of the honeymoon: reconstructing the Asian x European rose breeding history through the lens of genomics" by Leroy et al. provides an unprecedented insight into the genomic shifts that accompanied the breeding history of roses in Europe during the 19th century. By leveraging on genotyping and whole-genome sequencing data from more than 200 accessions, this study reconstructs the genetic journey of roses from their European origins to their integration with Asian varieties. This work is particularly fascinating for evolutionary biologists, geneticists, and horticulturists alike, as it sheds light on several critical aspects of plant breeding history. The study provides compelling evidence of a rapid transition from predominantly European to a near-Asian genetic background within a few generations. This shift highlights the strong influence of Asian roses in shaping modern cultivated varieties, particularly through the introduction of recurrent blooming traits, novel colors, and scents. A striking finding is the reduction in genetic diversity that occurred during this transition, likely due to selective breeding practices that prioritized a narrow set of desirable traits. This discovery underscores the potential risks of genetic bottlenecks in cultivated plants and raises concerns about the long-term sustainability of modern rose breeding programs. The study also features a robust methodological framework applied to a unique data set which includes 204 rose accessions, covering both botanically and historically significant varieties. Whole genome sequencing of 32 accessions provides high-resolution insights into genomic evolution. Comprehensive phenotypic characterization over multiple years further allows in-depth investigation of the genetic architecture of key horticultural traits such as petal number, flowering time, disease resistance, and scent composition, with the building of the largest GWAS catalog for roses to date. Finally, the findings emphasize the need for sustainable breeding practices that balance genetic innovation with the preservation of historical diversity, by maintaining rose collections. Integrating ancient genetic resources into modern breeding programs could enhance resilience against diseases and environmental changes, while maintaining the aesthetic qualities that make roses so beloved. Overall, the manuscript by Leroy et al. is a landmark contribution to our understanding of the genomic history of rose breeding. By merging historical records with cutting-edge genomic analysis, the study not only reconstructs a critical phase of horticultural evolution but also provides invaluable insights for future breeding strategies. References Thibault Leroy, Elise Albert, Tatiana Thouroude, Sylvie Baudino, Jean-Claude Caissard, Annie Chastellier, Jerome Chameau, Julien Jeauffre, Therese Loubert, Saretta Nindya Paramita, Alix Pernet, Vanessa Soufflet-Freslon, Cristiana Oghina-Pavie, Fabrice Foucher, Laurence Hibrand-Saint Oyant, Jeremy Clotault (2024) Dark side of the honeymoon: reconstructing the Asian x European rose breeding history through the lens of genomics. bioRxiv, ver.4 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.1101/2023.06.22.546162 | Dark side of the honeymoon: reconstructing the Asian x European rose breeding history through the lens of genomics | Thibault Leroy, Elise Albert, Tatiana Thouroude, Sylvie Baudino, Jean-Claude Caissard, Annie Chastellier, Jerome Chameau, Julien Jeauffre, Therese Loubert, Saretta Nindya Paramita, Alix Pernet, Vanessa Soufflet-Freslon, Cristiana Oghina-Pavie, Fab... | <p>Roses hold significant symbolic value in Western cultural heritage, often serving as a symbol of love and romance. Despite their ancient cultivation, the appreciation for the phenotypic diversity of roses emerged relatively recently, notably du... |  | Genotype-Phenotype, Hybridization / Introgression, Population Genetics / Genomics | mathieu gautier | Vincent Segura, Anonymous, Pierre Nouhaud | 2024-04-18 12:28:31 | View |
14 Jan 2025
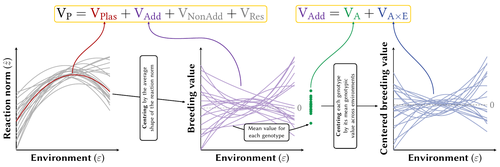
Partitioning the phenotypic and genetic variances of reaction normsPierre de Villemereuil, Luis-Miguel Chevin https://doi.org/10.32942/X2NC8BPhenotypic plasticity across traits, organisms and environments: decomposing the components of phenotypic varianceRecommended by Staffan Jacob based on reviews by Jarrod Hadfield and Thibaut Morel JournelPhenotypic plasticity can lead to rapid and important changes of trait distributions depending on environmental conditions, with important consequences for population dynamics, species interactions and adaptation. To better understand the evolution and importance of plasticity, we need to improve our ability to quantify and compare phenotypic plasticity among organisms, and estimate the evolutionary potential of plastic capacity. Plasticity is classically quantified through regression slopes, with units of traits per environment that by definition vary across organisms, traits and studies, and makes complex the comparison of how does plasticity vary across biological units, especially when linear versus quadratic reaction norms are considered. A clear methodology to quantify phenotypic plasticity in a way that allow for comparison across traits, organisms and environments is lacking. In this contribution, Pierre de Villemereuil and Luis-Miguel Chevin clarify key concepts about variability and evolutionary potential of plasticity, and propose an efficient method to partition phenotypic variance between genotype and environment. Expanding from the classical (too) simple regression slope approach, and directly integrating the genetic variability of plasticity, they provide a clear framework to quantify the part of phenotypic variance resulting from phenotypic plasticity, integrate the role of the shape of reaction norms, and estimate the heritable variation of trait plasticity. They integrate this method in a R package named Reacnorm, with step by step decision tree, that should greatly help the applicability of this framework. The authors here propose a contribution that simultaneously clarify key concepts and challenges about plasticity and reaction norms, being very interesting on the theoretical side, and is directly and quite simply applicable to any trait and organism. This should help and stimulate comparative studies of how does plasticity vary across traits, organisms and environmental contexts, both using already published datasets and through new experiments. References Pierre de Villemereuil, Luis-Miguel Chevin (2025) Partitioning the phenotypic and genetic variances of reaction norms. EcoEvorxiv, ver.4 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.32942/X2NC8B | Partitioning the phenotypic and genetic variances of reaction norms | Pierre de Villemereuil, Luis-Miguel Chevin | <p>Many traits show plastic phenotypic variation across environments, captured by their norms of reaction. These reaction norms may be discrete or continuous, and can substantially vary in shape across organisms and traits, making it difficult to ... |  | Evolutionary Ecology, Phenotypic Plasticity, Quantitative Genetics | Staffan Jacob | 2023-09-01 16:42:45 | View | |
03 Jan 2025
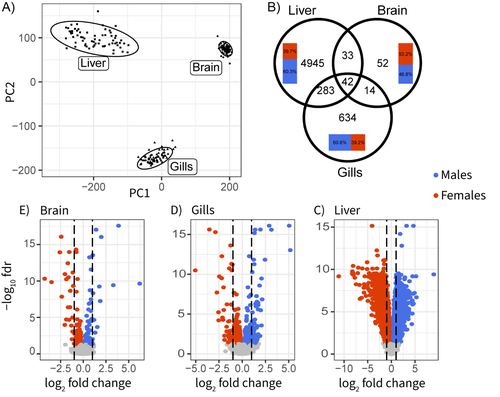
Sex-biased gene expression across tissues reveals unexpected differentiation in the gills of the threespine sticklebackFlorent Sylvestre, Nadia Aubin-Horth, Louis Bernatchez https://doi.org/10.1101/2024.06.09.597944Sex-biased gene expression in fish – a milestoneRecommended by Nicolas Galtier based on reviews by Qi Zhou and 2 anonymous reviewers based on reviews by Qi Zhou and 2 anonymous reviewers
There is a heavy body of literature on sex-biased gene expression, which can easily be tricky. One reason is that expression data are multi-dimensional, notoriously noisy, and highly sensitive to experimental conditions. Achieving reproducibility is, therefore, a challenge, especially in non-model organisms. Another reason is that the evolutionary forces shaping gene expression variation are complex, involving processes such as intra- and inter-locus conflicts between sexes, sex-chromosome evolution and degeneration, dosage compensation, cis vs. trans regulation, dominance, linkage, drift, etc... Not surprisingly, the field is rich in discordant studies, and a wide variety of situations have been described. The three-spined stickleback is a good illustration. This widely-studied fish displays conspicuous sexual dimorphisms, both morphological and behavioural. Yet, the existing literature regarding which genes and tissues are involved is remarkably unclear due to the lack of a comprehensive data set. Sylvestre et al. (1) here fix this issue. They sampled 40 wild-caught individuals in each of the two sexes and performed high-throughput sequencing of the transcriptome in three somatic tissues, dissected and preserved in the exact same conditions. This is an impressive effort, well above current standards. Data analysis delivered a series of neat results: gene expression in the liver is particularly sex-biased; the brain, in contrast, is remarkably little sex-differentiated despite the presence of courtship and paternal care in this species; gills show significantly sex-biased gene expression, which had been unnoticed previously despite the importance of this organ in fish ecotoxicology; the relatively young sex-chromosomes, finally, do not seem to experience dosage compensation, and are therefore enriched in sex-biased genes. Some of these results are consistent with previous studies in other fish species (2), here confirmed or demonstrated with a high degree of certainty. Others are new and worth considering in future studies of stickleback ecology and reproduction. We simply need more studies of this sort: well-conducted and clear-cut, recalling the career of its last author. References (1) Florent Sylvestre, Nadia Aubin-Horth, Louis Bernatchez (2024) Sex-biased gene expression across tissues reveals unexpected differentiation in the gills of the threespine stickleback. bioRxiv, ver.2 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.1101/2024.06.09.597944 (2) Iulia Darolti I, Judith E. Mank (2023). Sex-biased gene expression at single-cell resolution: cause and consequence of sexual dimorphism. Evolution Letters 7(3):148-156. https://doi.org/10.1093/evlett/qrad013
| Sex-biased gene expression across tissues reveals unexpected differentiation in the gills of the threespine stickleback | Florent Sylvestre, Nadia Aubin-Horth, Louis Bernatchez | <p>Sexual dimorphism can evolve through sex-specific regulation of the same gene set. However, sex chromosomes can also facilitate this by directly linking gene expression to sex. Moreover, differences in gene content between heteromorphic sex chr... |  | Expression Studies, Genetic conflicts, Genome Evolution, Molecular Evolution, Reproduction and Sex | Nicolas Galtier | 2024-06-11 18:57:06 | View | |
18 Dec 2024
Investigating the effects of diurnal and nocturnal pollinators on male and female reproductive success and on floral trait selection in Silene dioicaBarbot Estelle, Dufaÿ Mathilde, Godé Cécile, De Cauwer Isabelle https://doi.org/10.5281/zenodo.11488687More in less: almost everything you wanted to know about sex in flowers is in a single experiment with a single plant speciesRecommended by Juan Arroyo and Violeta Simón-Porcar and Violeta Simón-Porcar based on reviews by Luis Gimenez-Benavides, Andrea Cocucci, Giovanni Scopece and 1 anonymous reviewer based on reviews by Luis Gimenez-Benavides, Andrea Cocucci, Giovanni Scopece and 1 anonymous reviewer
Most flowering plants (almost 90% of species) are pollinated by animals (Ollerton et al. 2011). In fact, many plants are completely dependent on pollinator visits for reproductive success, due to the complete inability of selfing if they are self-incompatible or have strong gender differentiation, as in dioecious plants. Others have diminished reproductive output in the absence of pollinators, even being self-compatible, if their flowers present strong herkogamy or dichogamy, making autonomous selfing more difficult. Ultimately, all animal-pollinated plant species rely on pollinators for outcrossing. Depending on the genetic structure of plant populations and the movement patterns of these animals, outcrossing patterns will shape the population genetic variation, which will determine its adaptive fate. Thus, understanding the mechanisms governing the pollination interaction is crucial for unraveling the uncertainties of a huge proportion of biodiversity on Earth. Being mutualistic by definition, the animal side of this interaction is less understood, despite most pollinator groups being likely dependent on it for their persistence and perhaps diversity (Ollerton 2017). The role of pollinators in plant diversification has generated much literature and controversy ever since Darwin and his “abominable mystery” about angiosperm diversification (Friedman 2009). However, the other way around, that of plant`s effect on pollinator diversification, is more debatable. A remarkable example of this effect is the possible case of co-speciation mediated by nursery (brood site) pollination, which also includes antagonistic insect herbivory (Wiens et al. 2015), as in some Silene species and their moth pollinators and herbivores (Hembry and Althoff 2016). The authors of this recommendation benefitted from grants provided by grants PID2021-122715NB-I00 and TED2021-131037B-I00 funded by MCIN/AEI/ 10.13039/501100011033 and by the “European Union NextGeneration EU/PRTR”, and by MSCA-IF-2019-89789. Barbot, E., Dufaÿ, M., Godé, C., & De Cauwer, I. (2024). Exploring the effect of scent emission and exposition to diurnal versus nocturnal pollinators on selection patterns on floral traits. Zenodo. https://doi.org/10.5281/zenodo.11490231 Friedman, W. E. (2009). The meaning of Darwin's “abominable mystery”. American Journal of Botany, 96(1), 5-21. https://doi.org/10.3732/ajb.0800150 Haran, J., Kergoat, G. J., & de Medeiros, B. A. (2023). Most diverse, most neglected: weevils (Coleoptera: Curculionoidea) are ubiquitous specialized brood-site pollinators of tropical flora. Peer Community Journal, 3. https://doi.org/10.24072/pcjournal.279 Hembry, D.H. and Althoff, D.M. (2016), Diversification and coevolution in brood pollination mutualisms: Windows into the role of biotic interactions in generating biological diversity. American Journal of Botany, 103: 1783-1792. https://doi.org/10.3732/ajb.1600056 Kephart, S., Reynolds, R. J., Rutter, M. T., Fenster, C. B., & Dudash, M. R. (2006). Pollination and seed predation by moths on Silene and allied Caryophyllaceae: evaluating a model system to study the evolution of mutualisms. New Phytologist, 169(4), 667-680. https://doi.org/10.1111/j.1469-8137.2005.01619.x Kulbaba, M. W., & Worley, A. C. (2013). Selection on Polemonium brandegeei (Polemoniaceae) flowers under hummingbird pollination: in opposition, parallel, or independent of selection by hawkmoths?. Evolution, 67(8), 2194-2206. https://doi.org/10.1111/evo.12102 Nunes, C. E. P., Maruyama, P. K., Azevedo-Silva, M., & Sazima, M. (2018). Parasitoids turn herbivores into mutualists in a nursery system involving active pollination. Current Biology, 28(6), 980-986. https://doi.org/10.1016/j.cub.2018.02.013 Ollerton, J. (2017). Pollinator diversity: distribution, ecological function, and conservation. Annual review of ecology, evolution, and systematics, 48(1), 353-376. https://doi.org/10.1146/annurev-ecolsys-110316-022919 Ollerton, J., Winfree, R., & Tarrant, S. (2011). How many flowering plants are pollinated by animals?. Oikos, 120(3), 321-326. https://doi.org/10.1111/j.1600-0706.2010.18644.x Prieto-Benitez, S., Yela, J. L., & Gimenez-Benavides, L. (2017). Ten years of progress in the study of Hadena-Caryophyllaceae nursery pollination. A review in light of new Mediterranean data. Flora, 232, 63-72. https://doi.org/10.1016/j.flora.2017.02.004 Raguso, R. A. (2008). Wake up and smell the roses: the ecology and evolution of floral scent. Annual review of ecology, evolution, and systematics, 39(1), 549-569. https://doi.org/10.1146/annurev.ecolsys.38.091206.095601 Simón-Porcar, V. I., Meagher, T. R., & Arroyo, J. (2015). Disassortative mating prevails in style-dimorphic Narcissus papyraceus despite low reciprocity and compatibility of morphs. Evolution, 69(9), 2276-2288. https://doi.org/10.1111/evo.12731 Suetsugu, K. (2023). A novel nursery pollination system between a mycoheterotrophic orchid and mushroom-feeding flies. Ecology, 104(11), e4152. https://doi.org/10.1002/ecy.4152 Wiens, J. J., Lapoint, R. T., & Whiteman, N. K. (2015). Herbivory increases diversification across insect clades. Nature communications, 6(1), 8370. https://doi.org/10.1038/ncomms9370 | Investigating the effects of diurnal and nocturnal pollinators on male and female reproductive success and on floral trait selection in Silene dioica | Barbot Estelle, Dufaÿ Mathilde, Godé Cécile, De Cauwer Isabelle | <p>Plant species with mixed pollination systems are under pollinator-mediated selection by both diurnal and nocturnal pollinator species. This could impact the strength and potentially direction of selection on floral traits, as different pollinat... | Evolutionary Ecology, Reproduction and Sex | Juan Arroyo | 2024-06-05 15:52:46 | View | ||
18 Nov 2024

Faster model-based estimation of ancestry proportionsCindy G. Santander, Alba Refoyo Martinez, Jonas Meisner https://doi.org/10.1101/2024.07.08.602454fastmixture generates fast and accurate estimates of global ancestry proportions and ancestral allele frequenciesRecommended by Matteo Fumagalli based on reviews by Oscar Lao Grueso and 2 anonymous reviewers based on reviews by Oscar Lao Grueso and 2 anonymous reviewers
The estimation of ancestry proportions in individuals is an important analysis in both evolutionary biology and medical genetics. However, popular tools like ADMIXTURE (Alexander et al. 2009) and STRUCTURE (Pritchard et al. 2000) do not scale well with the large amount of data currently available. Recent alternative methods, such as SCOPE (Chiu et al. 2022), favour scalability over accuracy. In this study, Santander and coworkers introduce a new software, called fastmixture, which estimates ancestry proportions and ancestral allele frequencies using novel implementations for initialisation and convergence of its model-based algorithm (Santander et al. 2024). In simulated datasets, fastmixture displays desirable properties of speed and accuracy, with its performance surpassing commonly used software (Alexander et al. 2009, Pritchard et al. 2000, Chiu et al. 2022, Mantes et al. 2023). fastmixture is almost 30 times faster than ADMIXTURE under a complex model with five ancestral populations, while retaining similar accuracy levels. When applied to data from the 1000 Genomes Project (1000 Genomes Project Consortium 2025), fastmixture recapitulated expected levels of global ancestry. The new software is freely available on GitHub with an accessible documentation. fastmixture accepts input files in PLINK format. It remains an open question whether extensive parameter tuning could increase the scalability and accuracy of established methods. A comprehensive assessment of fastmixture over a wide range of data processing options (Hemstrom et al. 2024) is also missing. Finally, whether model-based approaches are fully scalable to ever increasing biobank datasets is still under debate. Nevertheless, the superior computational performance of fastmixture is evident and it is likely that this new software will soon replace existing popular tools to estimate global ancestry proportions. References Alexander DH, Novembre J, Lange K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 2009;19(9):1655-1664. https://doi.org/10.1101/gr.094052.109 Pritchard JK, Stephens M, Donnelly P. Inference of population structure using multilocus genotype data. Genetics. 2000;155(2):945-959. https://doi.org/10.1093/genetics/155.2.945 Chiu AM, Molloy EK, Tan Z, Talwalkar A, Sankararaman S. Inferring population structure in biobank-scale genomic data. Am J Hum Genet. 2022;109(4):727-737. https://doi.org/10.1016/j.ajhg.2022.02.015 Santander CG, Refoyo Martinez A, Meisner J. Faster model-based estimation of ancestry proportions. bioRxiv 2024; ver.2 peer-reviewed and recommended by PCI Evol Biol. https://doi.org/10.1101/2024.07.08.602454 Mantes AD, Montserrat DM, Bustamante CD, Giró-I-Nieto X, Ioannidis AG. Neural ADMIXTURE for rapid genomic clustering. Nat Comput Sci. 2023;3(7):621-629. https://doi.org/10.1038/s43588-023-00482-7 1000 Genomes Project Consortium, Auton A, Brooks LD, et al. A global reference for human genetic variation. Nature. 2015;526(7571):68-74. https://doi.org/10.1038/nature15393 Hemstrom W, Grummer JA, Luikart G, Christie MR. Next-generation data filtering in the genomics era. Nat Rev Genet. 2024;25(11):750-767. https://doi.org/10.1038/s41576-024-00738-6 | Faster model-based estimation of ancestry proportions | Cindy G. Santander, Alba Refoyo Martinez, Jonas Meisner | <p>Ancestry estimation from genotype data in unrelated individuals has become an essential tool in population and medical genetics to understand demographic population histories and to model or correct for population structure. The ADMIXTURE softw... |  | Bioinformatics & Computational Biology, Human Evolution, Population Genetics / Genomics | Matteo Fumagalli | 2024-07-14 11:48:39 | View | |
18 Nov 2024
A genomic duplication spanning multiple P450s contributes to insecticide resistance in the dengue mosquito Aedes aegyptiTiphaine Bacot, Chloe Haberkorn, Joseph Guilliet, Julien Cattel, Mary Kefi, Louis Nadalin, Jonathan Filee, Frederic Boyer, Thierry Gaude, Frederic Laporte, Jordan Tutagata, John Vontas, Isabelle Dusfour, Jean-Marc Bonneville, Jean-Philippe David https://doi.org/10.1101/2024.04.03.587871A duplication driving metabolic insecticide resistance in Aedes aegyptiRecommended by Diego A. Hartasánchez based on reviews by Diego Ayala and 1 anonymous reviewer based on reviews by Diego Ayala and 1 anonymous reviewer
Insecticide resistance in mosquitoes represents a notable challenge to public health efforts aimed at controlling vector-borne diseases. Among mosquito species, Aedes aegypti is particularly significant due to its extensive geographic spread and its ability to transmit arboviruses causing diseases such as dengue, yellow fever, Zika, and chikungunya (Brown et al., 2014). Insecticide resistance typically develops through two main mechanisms: target-site mutations, which affect the insecticide's interaction with its target, and metabolic resistance, in which insecticide detoxification is enhanced in mosquitoes. While target-site mutations are well characterized, the mechanisms underlying metabolic resistance are understudied. The study by Bacot and colleagues (2024) contributes to our understanding of the genetic and evolutionary mechanisms driving insecticide resistance, focusing on a case of metabolic resistance in Aedes aegypti from French Guiana. Following the recent identification of a copy number variant region on chromosome 1, potentially linked to overexpression of detoxification enzymes (Cattel et al., 2020), this study explores the region’s genomic architecture, its likely origin and provides compelling evidence for its role in insecticide resistance. Through RNA sequencing and whole-genome pool sequencing, the authors reveal that this 220 kilobase duplication increases the expression level of several clustered P450 genes. Cytochrome P450s are known to play a role in breaking down pyrethroids like deltamethrin, a commonly used insecticide. The role of P450 enzymes in detoxification was demonstrated by treating mosquitoes with piperonyl butoxide, a P450 enzyme inhibitor, and observing reduction in deltamethrin resistance, further confirmed by RNA interference experiments. Despite the clear advantages of this genomic duplication in conferring resistance, the study also uncovers a fitness cost associated with carrying the duplication. Through experimental evolution, the authors find that mosquitoes with the duplication experience reduced fitness in the absence of insecticide pressure. Given the regions structural complexity, the authors could not completely disassociate the effect of the duplicated region and that of a target-site mutation. However, they developed an assay that can accurately track the presence of this resistance allele in mosquito populations. Altogether, the study by Bacot et al. (2024) highlights the challenges of characterizing the phenotypic effect of copy number variant regions in complex genomes, such as that of Aedes aegypti. It emphasizes the need for further studies on the origin and spread of this duplication to better understand how similar resistance mechanisms might evolve and disseminate. Overall, the completeness and coherence of the narrative, the detailed and thorough analysis, and the insightful discussion, make this work not only a significant contribution to the field of insecticide resistance but an interesting read for the general evolutionary biology community. References Brown, J. E., Evans, B. R., Zheng, W., Obas, V., Barrera-Martinez, L., Egizi, A., Zhao, H., Caccone, A., & Powell, J. R. (2014). Human impacts have shaped historical and recent evolution in Aedes aegypti, the dengue and yellow fever mosquito. Evolution, 68(2), 514–525. https://doi.org/10.1111/evo.12281 Cattel, J., Faucon, F., Le Péron, B., Sherpa, S., Monchal, M., Grillet, L., Gaude, T., Laporte, F., Dusfour, I., Reynaud, S., & David, J. P. (2019). Combining genetic crosses and pool targeted DNA-seq for untangling genomic variations associated with resistance to multiple insecticides in the mosquito Aedes aegypti. Evolutionary applications, 13(2), 303–317. https://doi.org/10.1111/eva.12867 Tiphaine Bacot, Chloe Haberkorn, Joseph Guilliet, Julien Cattel, Mary Kefi, Louis Nadalin, Jonathan Filee, Frederic Boyer, Thierry Gaude, Frederic Laporte, Jordan Tutagata, John Vontas, Isabelle Dusfour, Jean-Marc Bonneville, Jean-Philippe David (2024) A genomic duplication spanning multiple P450s contributes to insecticide resistance in the dengue mosquito Aedes aegypti. bioRxiv, ver.5 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.1101/2024.04.03.587871 | A genomic duplication spanning multiple P450s contributes to insecticide resistance in the dengue mosquito *Aedes aegypti* | Tiphaine Bacot, Chloe Haberkorn, Joseph Guilliet, Julien Cattel, Mary Kefi, Louis Nadalin, Jonathan Filee, Frederic Boyer, Thierry Gaude, Frederic Laporte, Jordan Tutagata, John Vontas, Isabelle Dusfour, Jean-Marc Bonneville, Jean-Philippe David | <p>Resistance of mosquitoes to insecticides is one example of rapid adaptation to anthropogenic selection pressures having a strong impact on human health and activities. Target-site modification and increased insecticide detoxification are the tw... | Adaptation, Evolutionary Applications, Expression Studies, Genotype-Phenotype | Diego A. Hartasánchez | 2024-04-10 11:36:06 | View | ||
24 Oct 2024

Cryptic species and hybridisation in corals: challenges and opportunities for conservation and restorationCynthia Riginos, Iva Popovic, Zoe Meziere, Vhon Garcia, Ilha Byrne, Samantha Howitt, Hisatake Ishida, Kevin Bairos-Novak, Adriana Humanes, Hugo Scharfenstein, Thomas Richards, Ethan Briggs, Vanessa Clark, Chuan Lei, Mariam Khan, Katharine Prata https://doi.org/10.32942/X2502XHow common cryptic coral diversity can blur biodiversity metrics and challenge managementRecommended by Eric Pante based on reviews by 2 anonymous reviewers based on reviews by 2 anonymous reviewers
Biological conservation aims at protecting the genetic diversity generated by evolutionary processes over the course of life's history on earth (Allendorf and Luikart 2007), and to be effective, it requires that its fundamental units (among which populations and species) be delimited as precisely as possible. This exercise is particularly important for corals because hybridisation and introgression have played a fundamental role in shaping their contemporary diversity (eg Veron 1995). In their review paper, Riginos et al (2024) show that 68% of nominal taxa investigated for genomic population structure bear the molecular signature of partial reproductive isolation, and can be considered as cryptic genetic groups. Another review study (published a day before the preprint of Riginos et al), converges in the finding that cryptic diversity is rampant in nominal coral species (Grupstra et al 2024). This result has strong bearing on the study of coral biology; as Riginos et al state, "any coral investigation that does not genotype the corals under study risks treating a heterogeneous mix of partially reproductively isolated taxa as a single species." The stakes are therefore high, given the ecological importance of corals and the ecosystem services they provide. While Grupstra et al (2024) discusses the impact of cryptic coral diversity in the context of functional differences in thermal adaptation and the processes that lead to cryptic lineages, Riginos et al (2024) provide a quantitative review of cryptic lineages within nominal species, providing reproducible criteria for delineation, details the importance of detecting hybrids, summarises how biodiversity metrics and conservation efforts can be biased by unrecognised cryptic lineages, offer recommendations on how to recognise and deal with cryptic diversity, and discuss how corals can be regarded as highly valuable model systems to study adaptation and speciation. The study of Riginos et al (2024) is therefore a must-read to all coral biologists, especially those involved in biological conservation. References Fred W. Allendorf and Gordon Luikart (2007) Conservation and Genetics of Populations. Blackwell Publishing, Malden MA, USA. Carsten G. B. Grupstra, Matías Gómez-Corrales, James E. Fifer, Hannah E. Aichelman, Kirstin S. Meyer-Kaiser, Carlos Prada, Sarah W. Davies (2024) Integrating cryptic diversity into coral evolution, symbiosis and conservation. Nat Ecol Evol 8, 622–636 (2024). https://doi.org/10.1038/s41559-023-02319-y Cynthia Riginos, Iva Popovic, Zoe Meziere, Vhon Garcia, Ilha Byrne, Samantha Howitt, Hisatake Ishida, Kevin Bairos-Novak, Adriana Humanes, Hugo Scharfenstein, Thomas Richards, Ethan Briggs, Vanessa Clark, Chuan Lei, Mariam Khan, Katharine Prata (2024) Cryptic species and hybridisation in corals: challenges and opportunities for conservation and restoration. EcoEvoRxiv, ver.2 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.32942/X2502X J. E. N. Veron (1995) Corals in Space and Time: The Biogeography and Evolution of the Scleractinia. Cornell University Press | Cryptic species and hybridisation in corals: challenges and opportunities for conservation and restoration | Cynthia Riginos, Iva Popovic, Zoe Meziere, Vhon Garcia, Ilha Byrne, Samantha Howitt, Hisatake Ishida, Kevin Bairos-Novak, Adriana Humanes, Hugo Scharfenstein, Thomas Richards, Ethan Briggs, Vanessa Clark, Chuan Lei, Mariam Khan, Katharine Prata | <p style="text-align: justify;">The conservation and management of coral reef ecosystems will benefit from accurate assessments of reef-building coral species diversity. However, the true diversity of corals may be obfuscated by cryptic yet geneti... |  | Adaptation, Evolutionary Applications, Hybridization / Introgression, Population Genetics / Genomics, Speciation | Eric Pante | 2024-02-15 10:29:46 | View | |
26 Sep 2024
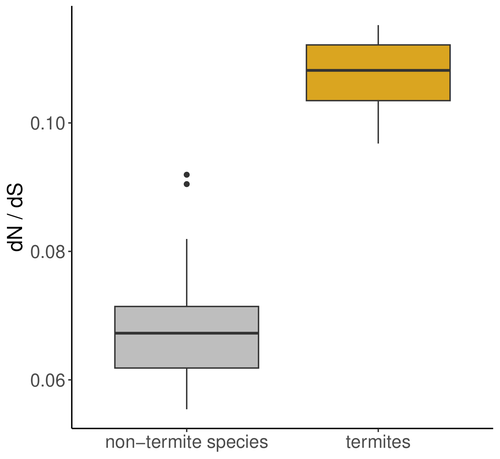
The impact of social complexity on the efficacy of natural selection in termitesCamille Roux, Alice Ha, Arthur Weyna, Morgan Lode, Jonathan Romiguier https://doi.org/10.1101/2024.04.26.591327Evolutionary trajectories of social transitions: Higher social complexity is associated with lower effective population size and reduced efficacy of selection in termitesRecommended by Trine Bilde based on reviews by 2 anonymous reviewers based on reviews by 2 anonymous reviewers
A comprehensive study by Roux et al 2024 investigates the impact of eusociality on the efficacy of natural selection in termites, with and additional focus on whether higher levels of social complexity are associated with lower effective population size (Ne) and relaxed purifying selection. Eusociality is characterized by a division of reproductive labor, cooperative care of offspring, and overlapping generations, and has evolved independently across various animal taxa with the most complex social systems found in Hymenoptera (bees, wasps, ants) and termites. Because reproduction is limited to a few individuals, this leads to a reduced effective population size (Ne), which impacts genome evolution. Smaller Ne increases the influence of genetic drift, weakening the efficiency of natural selection and allowing the accumulation of weakly deleterious mutations. This phenomenon, known as the "drift barrier," alters the mutation-selection balance in eusocial organisms. Studies in a range of social arthropods including ants, termites, crustaceans and spiders have shown an elevated ratio of nonsynonymous to synonymous substitutions (dN/dS), indicating relaxed purifying selection due to small Ne. In termite species with complex social structures, such as those with large colonies and high caste specialization, there are reports of higher dN/dS ratios compared to simpler social species. This suggests that higher social complexity, reflected in traits like nesting strategies and developmental pathways, further reduces Ne and the effectiveness of natural selection. The authors address these hypotheses by exploring the genomic impact of eusociality in termites (Isoptera) taking two approaches: First, they analyze transcriptome data from 66 Blattodea species and calculate the ratio of non-synonymous to synonymous mutations (dN/dS) as an indicator of natural selection efficiency and effective population size. They analyses reveal an increased dN/dS ratio in termites compared to other Blattodea species, reinforcing the notion that convergent evolution toward eusociality significantly reduces effective population size and weakens natural selection efficiency across the genome. Additionally, a comparison of 68 termite transcriptomes shows that this effect is more pronounced in species with higher social complexity. This is exciting as it advances our understanding of how increasing complexity in social organization decreases Ne and the efficiency of natural selection. The study substantiates the notion that social transitions follow evolutionary trajectories where lower and Ne and increasing drift have negative consequences for genome evolution (Ma et al 2024). References Camille Roux, Alice Ha, Arthur Weyna, Morgan Lode, Jonathan Romiguier (2024) The impact of social complexity on the efficacy of natural selection in termites. bioRxiv, ver.2 peer-reviewed and recommended by PCI Evol Biol. https://doi.org/10.1101/2024.04.26.591327 Jilong Ma, Jesper Bechsgaard, Anne Aagaard, Palle Villesen, Trine Bilde, Mikkel Heide Schierup (2024) Sociality in spiders is an evolutionary dead-end. | The impact of social complexity on the efficacy of natural selection in termites | Camille Roux, Alice Ha, Arthur Weyna, Morgan Lode, Jonathan Romiguier | <p style="text-align: justify;">In eusocial species, reproduction is monopolized by a few reproductive individuals. From the perspective of population genetics, this implies that the effective population size (Ne<em>)</em> of these organisms is li... |  | Molecular Evolution | Trine Bilde | 2024-04-30 12:10:20 | View | |
24 Sep 2024
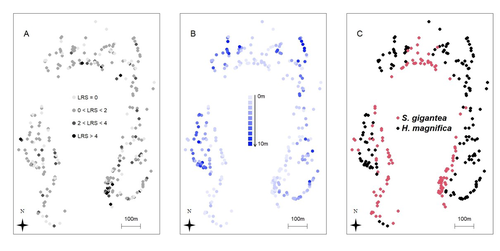
Spatial autocorrelation and host anemone species drive variation in local components of fitness in a wild clownfish populationPascal Marrot, Cécile Fauvelot, Michael L. Berumen, Maya Srinivasan, Geoffrey P. Jones, Serge Planes, and Benoit Pujol https://doi.org/10.5281/zenodo.13806778Is our best measure of fitness correlated with environment? A study in an orange clownfish population.Recommended by Pierre de Villemereuil based on reviews by Stefan Vriend and 2 anonymous reviewers based on reviews by Stefan Vriend and 2 anonymous reviewers
Getting a clear definition of fitness for a particular evolutionary biology question is a complex challenge, fraught with pitfalls and misconceptions (Orr, 2009; Walsh & Lynch, 2018). In longitudinal surveys of wild populations, lifetime reproductive success (LRS) is generally considered the best measure of individual fitness (Bonnet, 2022). However, it is important to bear in mind that LRS is only a (noisy) measure of the realised success, relying on a substantial amount of assumptions (e.g. with regard to generation overlap, Walsh & Lynch, 2018), not a direct measure of fitness. In a study on the clownfish, Marrot et al. (2024) studied the spatial and ecological drivers of lifetime reproductive success. To do so, they analysed a 10-year long survey on over 300 anemones harbouring clownfishes, and used a genetics-based pedigree to infer the LRS of each individual. Using a characterisation of the micro-habitat provided by each anemone, they used the anemone species, density and depth as ecological drivers and spatial-autocorrelated models to study more general (and undefined) spatial drivers. The authors found that LRS was influenced by a significant amount by the spatial structure of the population, and, to some extent, by the anemone species harbouring the clownfish individuals. Together, they explain a substantial proportion of the individual variation in LRS. While the actual determinants of spatial variation of LRS in this (and other) species remain understood, this study highlights an important aspect of measuring fitness in wild populations using LRS: it is particularly noisy and subject to environmental variation. This certainly does not mean that LRS is a bad proxy for fitness, it is still among the best measure of it we can have access to. However, it highlights how carefully we should thread when analysing it. Especially, spatial auto-correlation of LRS, combined with population structure within a population, would lead to genotype-environment correlation for fitness, which is likely to bias predictions of response to natural selection and would be extremely difficult to estimate (Falconer & Mackay, 1996). References Pascal Marrot, Cécile Fauvelot, Michael L. Berumen, Maya Srinivasan, Geoffrey P. Jones, Serge Planes, and Benoit Pujol (2024) Spatial autocorrelation and host anemone species drive variation in local components of fitness in a wild clownfish population. Zenodo, ver.3 peer-reviewed and recommended by PCI Evol Biol https://doi.org/10.5281/zenodo.13806778 Bonnet, T., Morrissey, M. B., de Villemereuil, P., Alberts, S. C., Arcese, P., Bailey, L. D., Boutin, S., Brekke, P., Brent, L. J. N., Camenisch, G., Charmantier, A., Clutton-Brock, T. H., Cockburn, A., Coltman, D. W., Courtiol, A., Davidian, E., Evans, S. R., Ewen, J. G., Festa-Bianchet, M., … Kruuk, L. E. B. (2022). Genetic variance in fitness indicates rapid contemporary adaptive evolution in wild animals. Science, 376(6596), 1012–1016. https://doi.org/10.1126/science.abk0853 Falconer, D. S. and Mackay, T. F. C. (1996). Introduction to quantitative genetics (4th ed.). Benjamin Cummings. Orr, H. A. (2009). Fitness and its role in evolutionary genetics. Nature Reviews Genetics, 10(8), 531–539. https://doi.org/10.1038/nrg2603 Walsh, B. and Lynch, M. (2018). Evolution and selection of quantitative traits. Oxford University Press. | Spatial autocorrelation and host anemone species drive variation in local components of fitness in a wild clownfish population | Pascal Marrot, Cécile Fauvelot, Michael L. Berumen, Maya Srinivasan, Geoffrey P. Jones, Serge Planes, and Benoit Pujol | <p style="text-align: justify;">The susceptibility of species to habitat changes depends on which ecological drivers shape individual fitness components. To date, only a few studies have quantified fitness components such as the Lifetime Reproduct... |  | Adaptation, Evolutionary Ecology, Quantitative Genetics | Pierre de Villemereuil | 2023-07-31 11:42:58 | View |
FOLLOW US
MANAGING BOARD
Dustin Brisson
Julien Dutheil
Marianne Elias
Inês Fragata
François Rousset
Sishuo Wang










